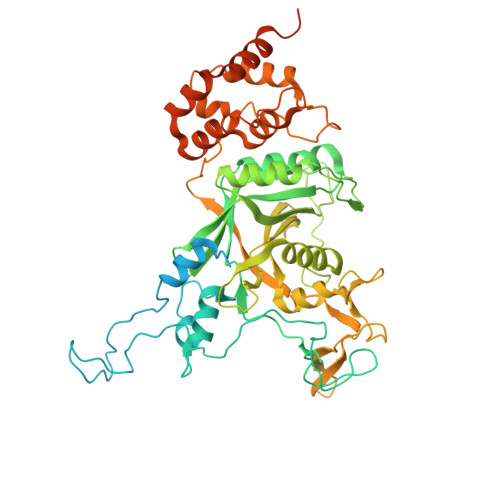
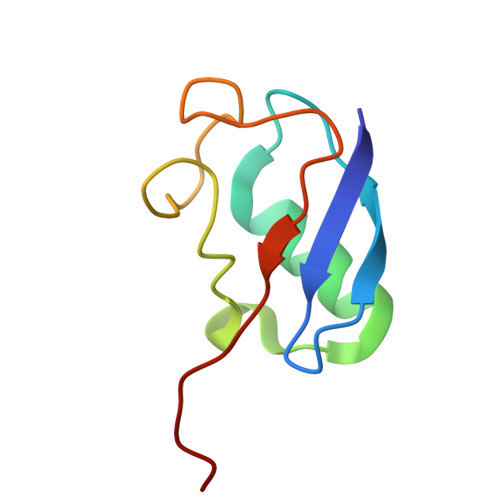
Faf1 accelerates p97-mediated protein unfolding by promoting ubiquitin engagement.
Liao, Z., Arkinson, C., Martin, A.(2025) bioRxiv
- PubMed: 41278724
- DOI: https://doi.org/10.1101/2025.10.27.684972
- Primary Citation Related Structures:
11SY, 11TA, 11VE - PubMed Abstract:
P97/VCP is a protein unfoldase of the AAA+ ATPase family that plays essential roles in numerous cellular processes, including ER-associated degradation and DNA replication. P97 utilizes various cofactors to process different substrates. For unfolding of proteins that are modified with K48-linked ubiquitin chains, p97 works with the heterodimeric cofactor Ufd1-Npl4, and the cofactor Faf1 was shown to enhance this activity in the context of replisome disassembly, yet the underlying mechanisms remain unknown. Here, we employ an in vitro reconstituted system with human components for biochemical experiments, mutational studies, FRET-based assays, and cryo-EM structure determination to reveal that Faf1 plays a generic role in accelerating ubiquitin-dependent substrate processing by promoting the unfolding of an initiator ubiquitin and its engagement by the ATPase motor. Faf1 thereby uses its p97-bound C-terminal UBX domain to anchor a long helix that braces the UT3 domain of Ufd1 and apparently stabilizes the Ufd1-Npl4 cofactor for ubiquitin unfolding. Our findings demonstrate how p97 works simultaneously with several cofactors to facilitate the unfolding of ubiquitinated proteins, indicating more complex regulatory mechanisms for substrate selection than for the simpler Cdc48 ortholog in yeast.
- California Institute for Quantitative Biosciences, University of California at Berkeley, Berkeley, CA 94720, USA.
Organizational Affiliation: